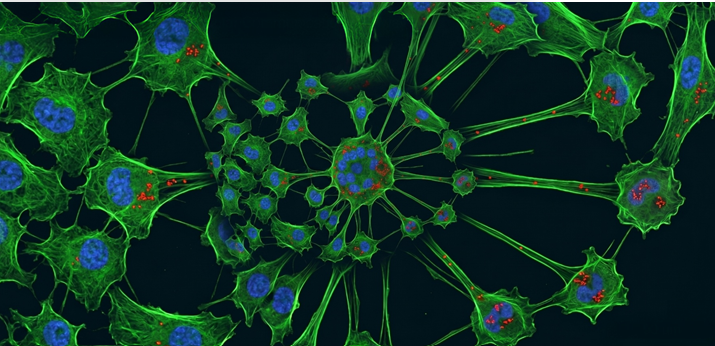
To understand neurodegenerative diseases like ALS, researchers are looking beyond individual neurons and examining tiny bridges that connect neurons to other neurons.
These bridges, called tunneling nanotubes (TNTs), shuttle molecules and proteins between cells, including small structures with specific functions called organelles. But many questions remain about just how these bridges form and what different proteins and signals they might transfer.
 At the Les Turner ALS Center at Northwestern Medicine, scientist Yvette Wong is using high-tech microscopy to better understand these bridges—and find out if and how they might be implicated in ALS.
At the Les Turner ALS Center at Northwestern Medicine, scientist Yvette Wong is using high-tech microscopy to better understand these bridges—and find out if and how they might be implicated in ALS.
“This is a really new area of research,” said Wong, assistant professor of neurology at Northwestern’s Feinberg School of Medicine. “There has been little research in whether and how TNTs might be important in ALS. But other research has shown they could be important in diseases like Parkinson’s, so we think it’s an area to learn more.”
Wong uses super-resolution microscopy techniques that can zoom in to just 100 microns wide—many times smaller than one cell—to understand how TNTs grow. Her lab uses different colored fluorescent tags to track components in a petri dish, including neurons and proteins, then tracks those components as they change over time.
“We can take really short, fast movies, like every second, or we can take a snapshot of every ten minutes for 24 hours,” she said. “That way, we can watch how these dynamics change on different time scales.”
With funding from the Les Turner ALS Foundation, Wong’s team mixed different cell types to find out how to get TNTs to grow in the dish. “They are not abundant in every cell type, and they may only form in certain conditions, so we needed to figure out how to better make them,” Wong said.
Then, they figured out a method to understand what TNTs transfer between cells. Once they started visualizing the dynamics of organelles and mitochondria—the powerhouses of cells—in TNTs, they knew they were on the right track. “It’s been really exciting to be able to visualize this,” she said.
Now, the lab’s goal is to try to track whether TNTs are involved in regulating important proteins like TDP-43, which is a hallmark of ALS. The majority of ALS cases show an unusual aggregation of this protein, and it’s believed that this dysfunction leads to neurodegeneration.
“Does the TNT transfer TDP-43 or perhaps transfer other signals which can regulate TDP-43 dynamics in neurons? And if so, is that a normal way for cells to communicate with each other, and does it become dysfunctional in ALS?” she said. “If we find out that these TNTs are one way to regulate this toxic protein, there may be a way to modulate that signal.”
“The work by the Wong lab addresses an important question in the field—how does pathology spread among components of a network of connected neurons?” said Robert Kalb, director of the Les Turner ALS Center at Northwestern Medicine. “The nanotube idea is compelling, and this new work provides evidence that they could be relevant to human disease.”
The team is also using this microscopy tool to study protein dynamics in Parkinson’s disease. “There are so many parallels across neurological diseases, that when we learn about dynamics in one disease, it helps us think about the other,” she said.
Funding from the Les Turner ALS Foundation helped get the TNT project off the ground and will help Wong’s lab be competitive for larger grants from the National Institutes of Health.
“Because it’s a relatively new field, it doesn’t have a long track record of data, so we need funding to help us create that data,” she said. “We think TNTs may help us better understand the possible steps contributing to ALS, so this funding has been really helpful for us to start asking basic questions about TNT transfer and protein dynamics.”